(Le communiqué est en anglais.)
Kannalife, Inc. (OTC: KLFE), a biopharmaceutical and phyto-medical company innovating cannabinoid therapeutics, has selected Padilla as a strategic partner for its public relations and communications programs.
Headquartered in Doylestown, Pa., Kannalife has extensively researched and improved cannabidiol (CBD) by using it to create new chemical entities to treat painful neurodegenerative and oxidative stress-related diseases.
Padilla will execute a strategic communications initiative to help elevate awareness and appreciation for Kannalife’s scientific platform and its innovative approach to cannabinoid therapeutics and CBD-related drug discovery. Padilla will focus its efforts on media relations, thought leadership and social media strategy to position Kannalife as a leader in cannabinoid therapeutics.
“We’re thrilled to be helping Kannalife establish its position and point of view within the exciting cannabinoid therapeutics space,” said Matt Kucharski, Padilla’s president. “The company’s intellectual property and experienced management team make it a dynamite entrant in an important emerging market.”
Kannalife has been the only commercial drug discovery company in the cannabinoid therapeutics space to license the ‘507 Patent from the National Institutes of Health. Over the past decade, Kannalife has made significant improvements in the chemistry and efficacy of CBD-like molecules that are up to 200 times more potent, 10 times more bioavailable, 5 times safer, and 1,000 times more effective (therapeutic index) than CBD.
“Kannalife is at the center of some of the largest social and public health issues today: the unregulated use of CBD as an all-purpose medicine and the overuse of opioids for pain management in the U.S. and around the world. With the help of Padilla, we are well positioned to grow as the leader in cannabinoid therapeutics and CBD-related research and development,” said Dean Petkanas, Kannalife’s CEO.
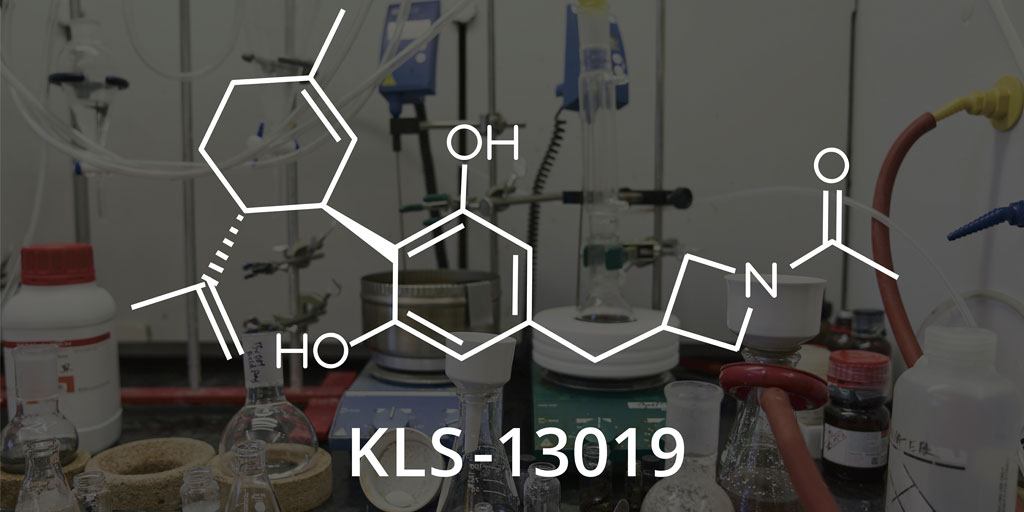
Kannalife’s Chief Scientific Officer, Bill Kinney, added, “Through a National Institute of Health/National Institute on Drug Abuse-funded Phase 1 STTR grant, we’ve determined that our cannabidiol-inspired, new chemical entity KLS-13019 has very high potential as a non-opioid, non-addictive compound. This molecule – we like to call CBD 2.0 – is a neuroprotectant, and has the potential to prevent and reverse neuropathic pain.”
Padilla will help accomplish the company’s communications initiative with a team that includes a blend of experts in health, corporate communications, media relations and social media.
About Padilla
Padilla is an independently operated, globally resourced public relations and communication company with offices across the United States. The agency builds, grows and protects brands and reputations worldwide by creating purposeful connections with the people who matter most through public relations, advertising, digital and social marketing, investor relations and brand strategy. Padilla includes the brand consultancy of Joe Smith, the food and nutrition experts at FoodMinds and the research authorities at SMS Research Advisors. Padilla’s clients include 3M, Blue Cross and Blue Shield of Minnesota, Cargill Animal Nutrition, Hass Avocado Board, Mayo Clinic, Prosciutto di Parma, Rockwell Automation, Sanofi Pasteur, U.S. Highbush Blueberry Council and Welch’s. Padilla is an AVENIR GLOBAL company and a founding member of the Worldcom Public Relations Group, a partnership of 132 independently owned partner offices in 115 cities on six continents. Connect with purpose at PadillaCo.com.
About Kannalife, Inc.
Kannalife, Inc., is a biopharmaceutical company leading innovation in research, development, and discovery of natural and novel biomimetic cannabinoid therapeutic agents. The company is focused on the development of proprietary cannabidiol (CBD) and CBD-like molecules for patients suffering from unmet medical needs of neurodegenerative disorders – including chemotherapy-induced peripheral neuropathy (CIPN), a chronic neuropathy caused by toxic chemotherapeutic agents; hepatic encephalopathy (HE), a neurotoxic brain-liver disorder caused by excessive concentrations of ammonia and ethanol in the brain; mild traumatic brain injury (mTBI), a disorder associated with single and repetitive impact injuries; and chronic traumatic encephalopathy (CTE), a disease associated with highly repetitive impact injuries in professional and amateur sports.
The company’s family of proprietary molecules focuses on treating oxidative stress-related diseases such as HE, chronic pain from neuropathies like CIPN and neurodegenerative diseases like CTE. Kannalife conducts its research and development efforts at the Pennsylvania Biotechnology Center of Bucks County in Doylestown, Pa.
For more information about Kannalife, Inc., visit Kannalife.com.
Forward-Looking Statements
This press release may contain certain forward-looking statements and information, as defined within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934 and is subject to the Safe Harbor created by those sections. This press release contains statements about expected future events, the company’s business plan, plan of operations, the viability of the company’s drug candidates, and/or financial results that are forward-looking in nature and subject to risks and uncertainties. Such forward-looking statements, by definition, involve risks and uncertainties. The company does not sell or distribute any products that are in violation of the United States Controlled Substances Act.
FDA Disclaimer
The statements made in this press release have not been evaluated by the Food and Drug Administration (FDA). These statements and the drug candidates of the company are not intended to diagnose, treat or cure any disease. The efficacy of these drug candidates have not been confirmed by FDA-approved research.
